
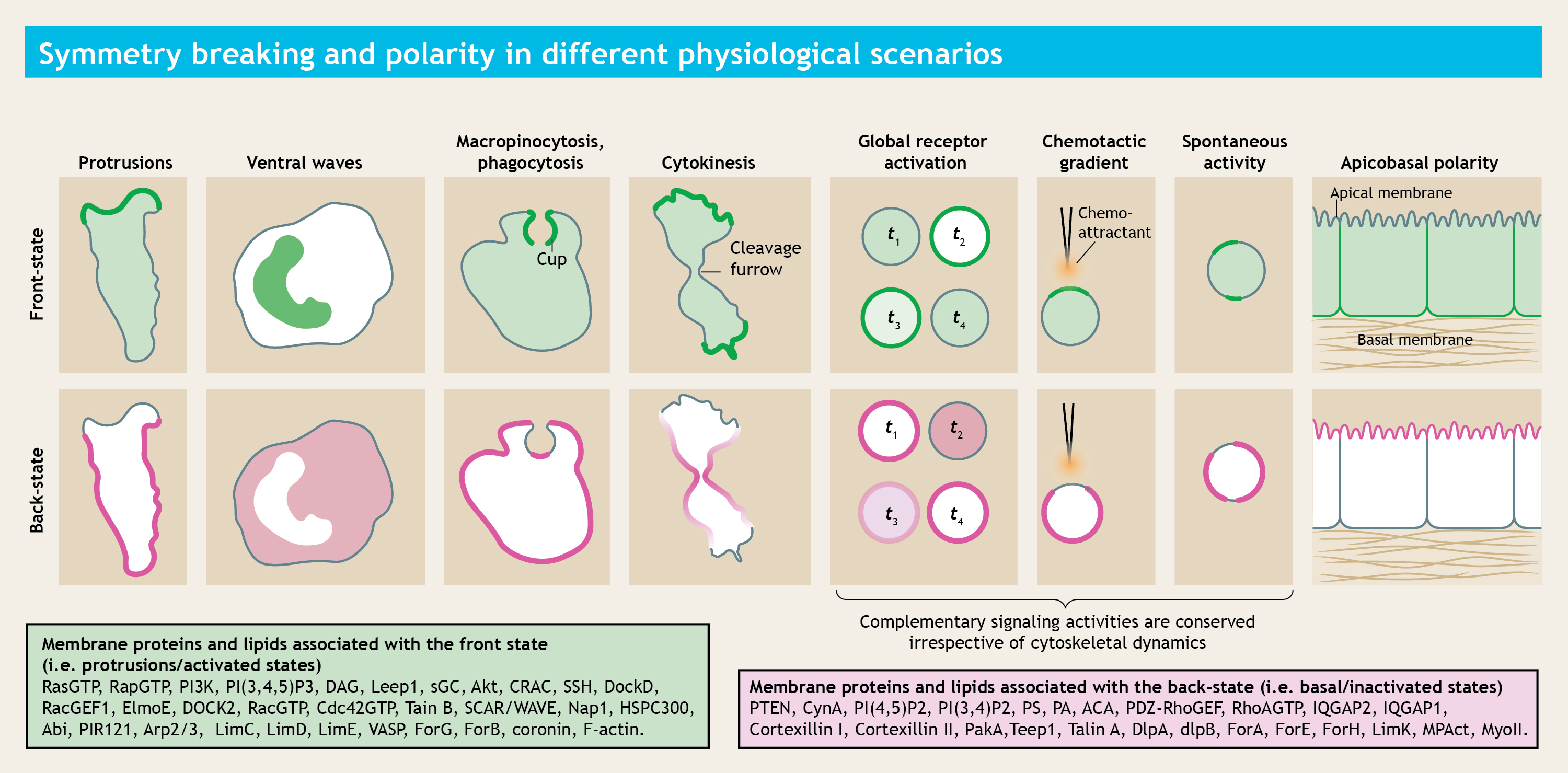
During diverse physiological scenarios, the plasma membrane undergoes symmetry transitions to dynamically create "front/activated" and "back/inactivated/basal" state regions. While a comprehensive catalog of specific proteins and lipids that asymmetrically localize to these regions has been compiled, many fundamental questions remained unanswered.
How does the symmetry breaking process is triggered in the plasma membrane, even before cell undergoes any mechanical deformation? What defines the state of a membrane domain? How different proteins and lipids self-organize themselves into these segregated membrane domains?
I have a long-standing research goal of understanding complex biomolecular phenomena using ab initio physicochemical principles. During my PhD, I was broadly interested in understanding how a cell continuously decide when and where to make next protrusions and contractions, in order to correctly migrate directionally under external cues or randomly under stochastic fluctuations.
Looking for a quick overview?
To get a general idea of our overall research area, please read our review paper.
We have used multimodal live-cell imaging techniques such as confocal microscopy (including super-resolution microscopy and photoconversion microscopy) and TIRF microscopy (including single-molecule measurements), engineered new optogenetic and synthetic perturbation tools, and developed new computational models to focus on three primary questions:

A schematic showing how the surface charge of the inner leaflet of the plasma membrane defines the state of signal transduction and cytoskeletal networks.
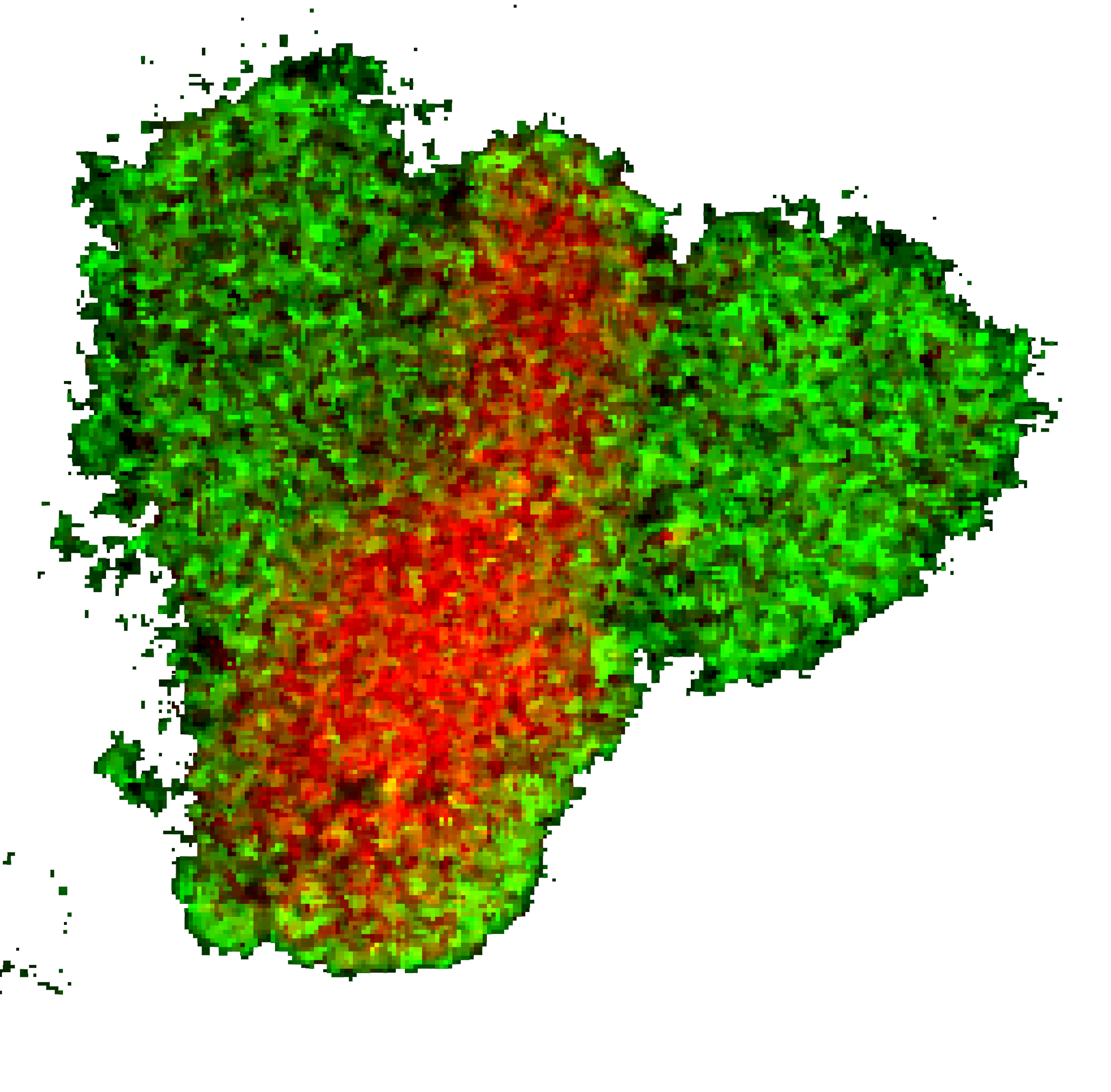
We demonstrated that the dynamic regulation of inner membrane surface charge can define the state of a membrane domain and thereby regulate the cell polarity and migration mode. We developed novel in situ monitoring tools and optogenetic actuators that can work in conjunction with standard live-cell imaging setup and genetic/pharmacological perturbations. Using these systems, we established that surface potential on the inner leaflet of the plasma membrane, a biophysical property — not some coincidental congruence of stepwise specific biomolecular interactions — spatially and temporally orchestrate Ras/PI3K/mTORC2/Akt/F-actin signaling activities in the cell to control protrusion formation. Our experiments demonstrated that surface charge is dynamically altered during signaling network activation and, in turn, its generic perturbation can induce or inhibit signaling activities that mediate cell migration.
It is well known that during propagation of nerve impulse, transmembrane potential can regulate the opening of the specific ion channels, which in turn collectively define the transmembrane potential. Our results indicated that transiently lowered inner membrane surface potential, which we termed “action surface potential”, can analogously propagate and interact with signaling network activation.
To learn in more detail, please read this paper.

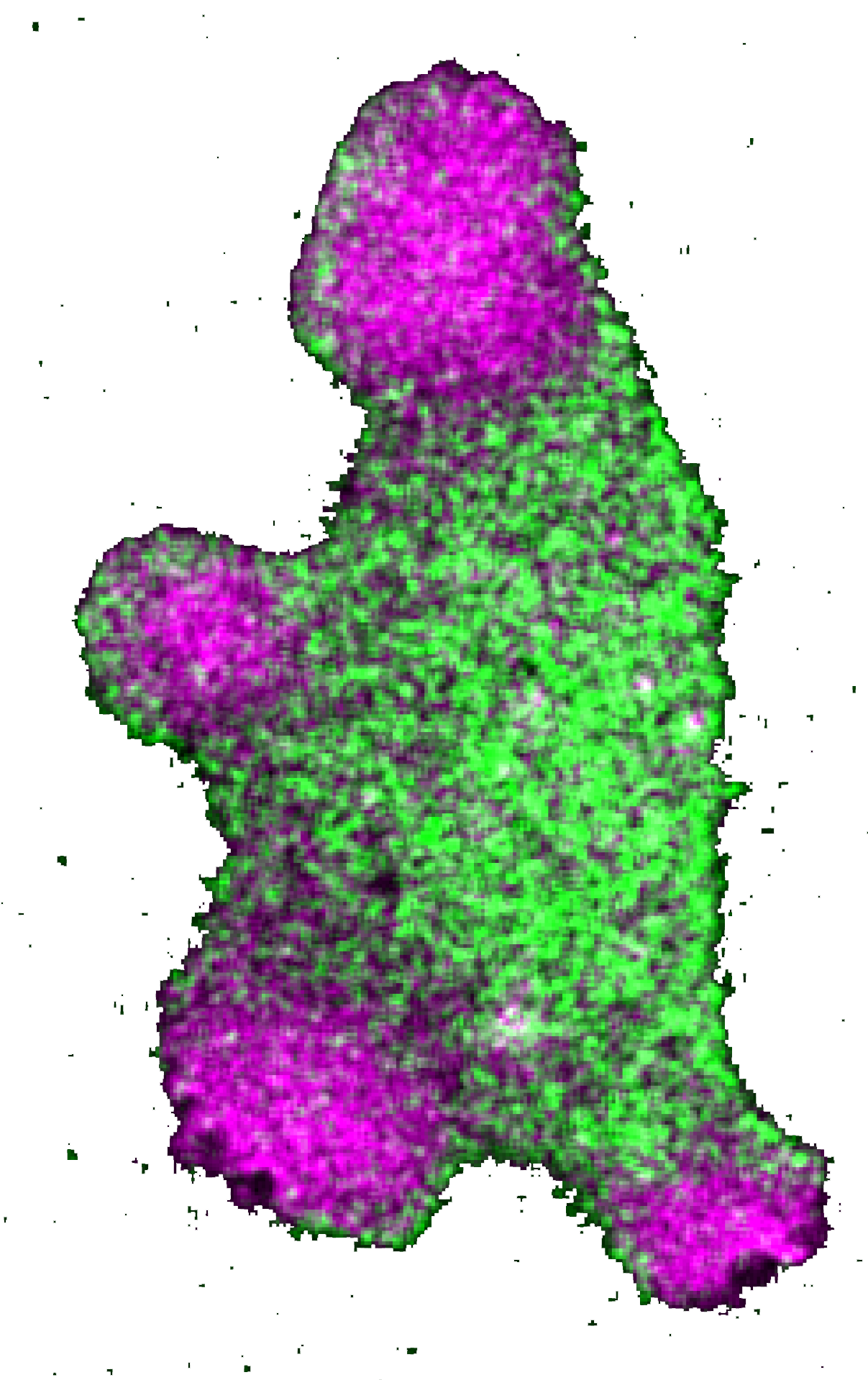
A schematic showing how the lipid-anchored proteins can dynamically partition into different membrane domains, based on their spatially heterogeneous mobility/diffusion rates.
This study began from a series of counterintuitive observations where we found that multiple lipid-anchored proteins, which were reported in the earlier literature to be uniform on the membrane, exhibited dynamic symmetry breaking in different cell physiological processes. Through a series of multimodal live-cell imaging, receptor activation assays, photoconversion microscopy, optogenetics, and in situ single-molecule measurements, and stochastic computational simulations, we established the principle of a unique dynamic partitioning mechanism. As we attributed the altered diffusion rates to altered binding affinity towards different domains of the membrane, we demonstrated that the spatially heterogenous mobility-based dynamic partitioning is sufficient to bring about familiar polarized pattern on the membrane.
Our proposed mechanism essentially serves as a distinct, energy-efficient alternative to conventional vesicular trafficking or picket-fence or other cytoskeletal restriction-based compartmentalization mechanisms. Since dynamic partitioning depends only on the inherent biophysical properties of the membrane and the affinity of the protein of interest, we believe, in future, this could emerge as one of the fundamental organizing principles in different biological processes where, either spontaneously or under the effect of external cues, plasma membrane polarization happens and different classes of membrane proteins get compartmentalized into specific domains (please see the first figure above for examples).
To learn in more detail, please read this paper.
=== This section maybe updated in the future. ===
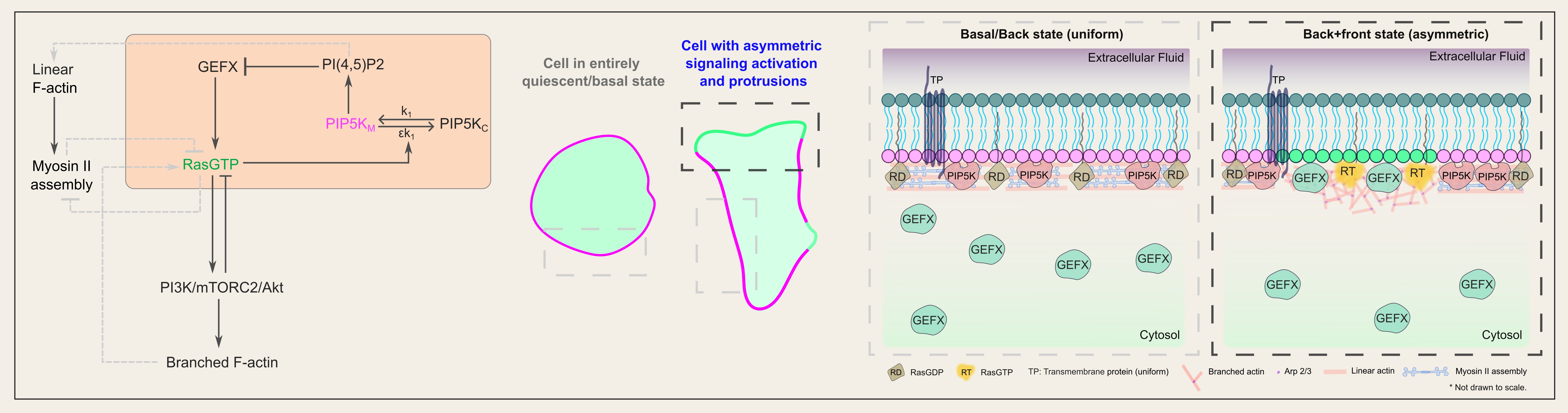
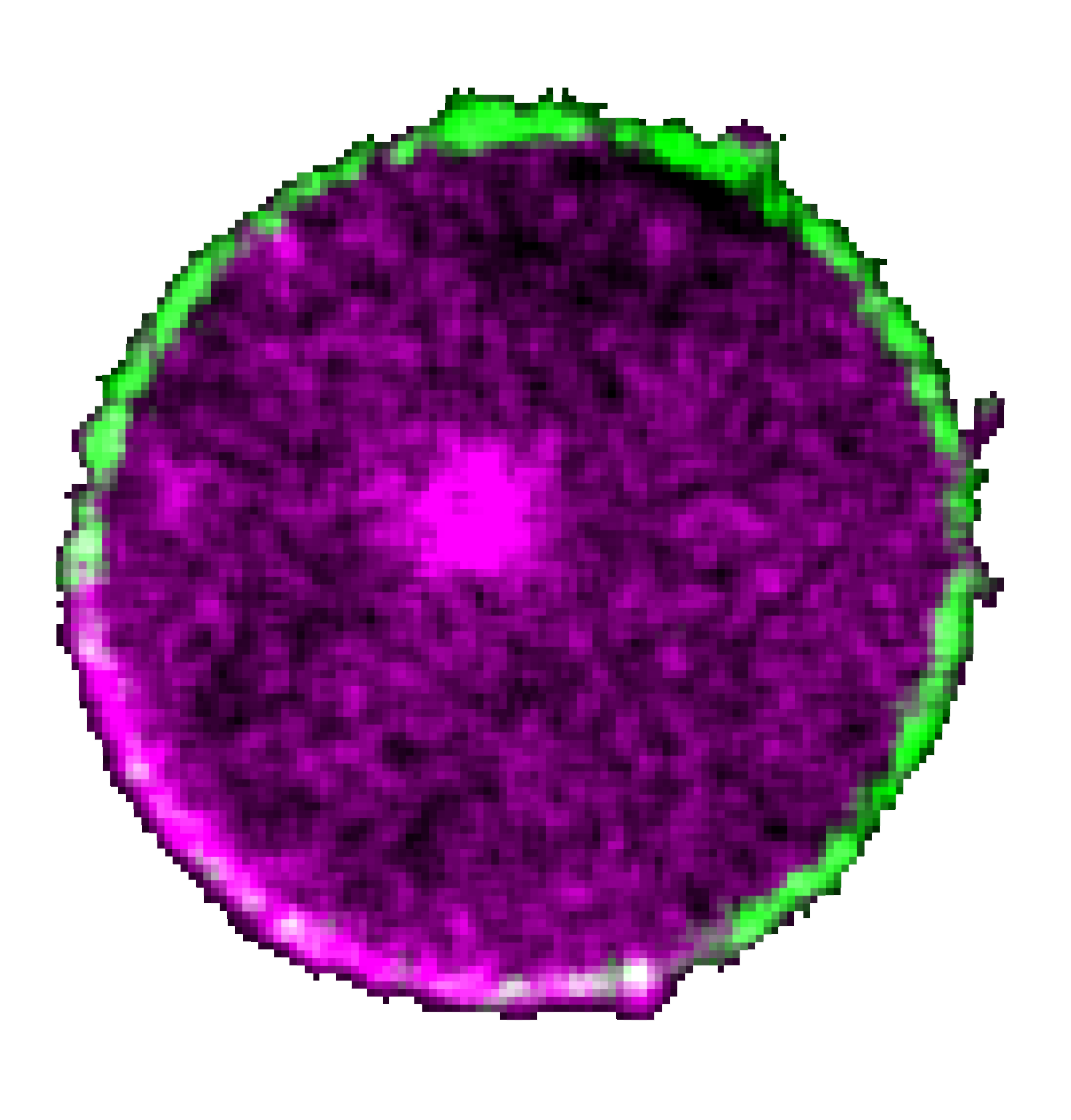
A schematic showing a bistable circuit of PIP5K and activated Ras protein can trigger symmetry breaking and tightly regulate cell polarization during migration.
Recent studies demonstrated that local activation of Ras can trigger F-actin-rich protrusions. However, to properly migrate and polarize, a cell must activate Ras in a particular domain on the plasma membrane, self-amplify that signal, and, at the same time, prevent the signal from initiating elsewhere. While multiple network topologies and biophysical mechanisms have been theorized, the mechanisms that bring about such symmetry breaking and cell polarization, which occur even in the absence of cytoskeletal dynamics and receptor inputs, have yet to be discovered. We addressed this question by showing that PIP5K, the kinase that converts PI4P to PI(4,5)P2, is the master regulator of the initial step of membrane symmetry breaking that leads to local Ras activation, downstream PI3K/Akt/Rac/Arp2/3 signaling activities, and the equilibrium between branched and linear actin polymerization. As we engineered novel optogenetic actuators to acutely alter membrane PIP5K level, we discovered that tuning the strength of feedback loops in the PIP5K/GTP-bound Ras bistable circuit is necessary and sufficient to trigger symmetry breaking as well as to induce a broad spectrum of cellular morphologies and phenotypes, underscoring the role of the circuit in controlling cell migration within a nonlinear dynamic continuum.
Combining data from single-molecule measurements of PIP5K diffusion and dissociation dynamics, a genetic screening for Ras activators, and biochemical/mechanical computational simulations, we identified a unique cell-intrinsic biophysical/molecular mechanism that we named high-affinity-driven immediate shuttling, by which activated Ras and PIP5K spatiotemporally limit each other’s activities and thereby completely define the membrane states and cell polarity during migration.
To learn in more detail, please read this paper.
New Software!
Recently, we have developed a new cross-platform, free and open-source, GUI software for automated generation and analysis of kymographs along dynamic boundaries, by implementing a custom rotational offset minimization algorithm. If you are interested in generating kymographs across some membrane/cortex of a cell that rapidly changes its shape, please check this GitHub repository and this paper (experience with programming or dependency management is unnecessary!).
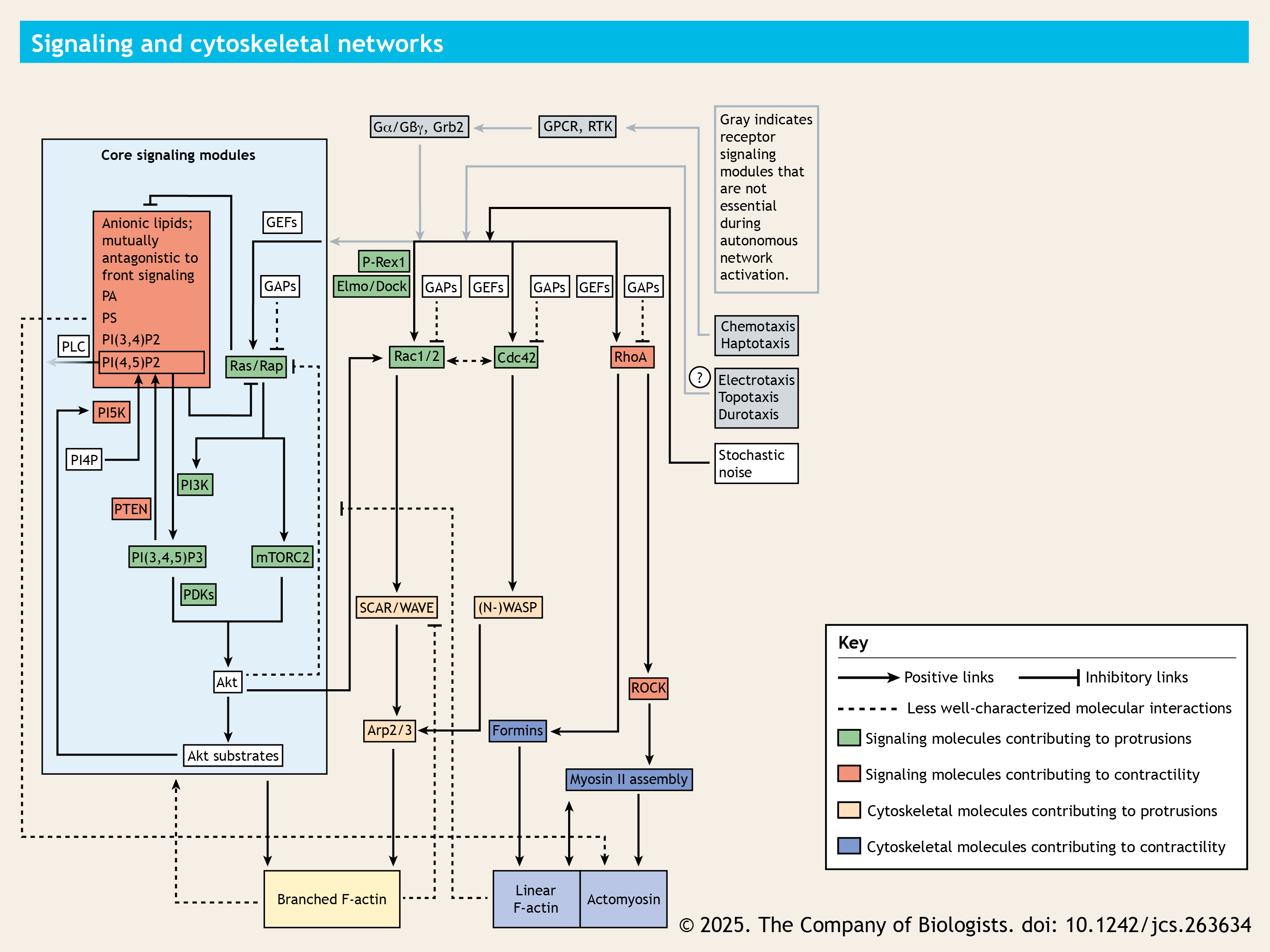
A few selected important signaling and cytoskeletal network components and their interactions are shown here. This network is largely conserved and regulates the migration, from single-celled amoeba Dictyostelium discoideum to human neutrophils and macrophages.
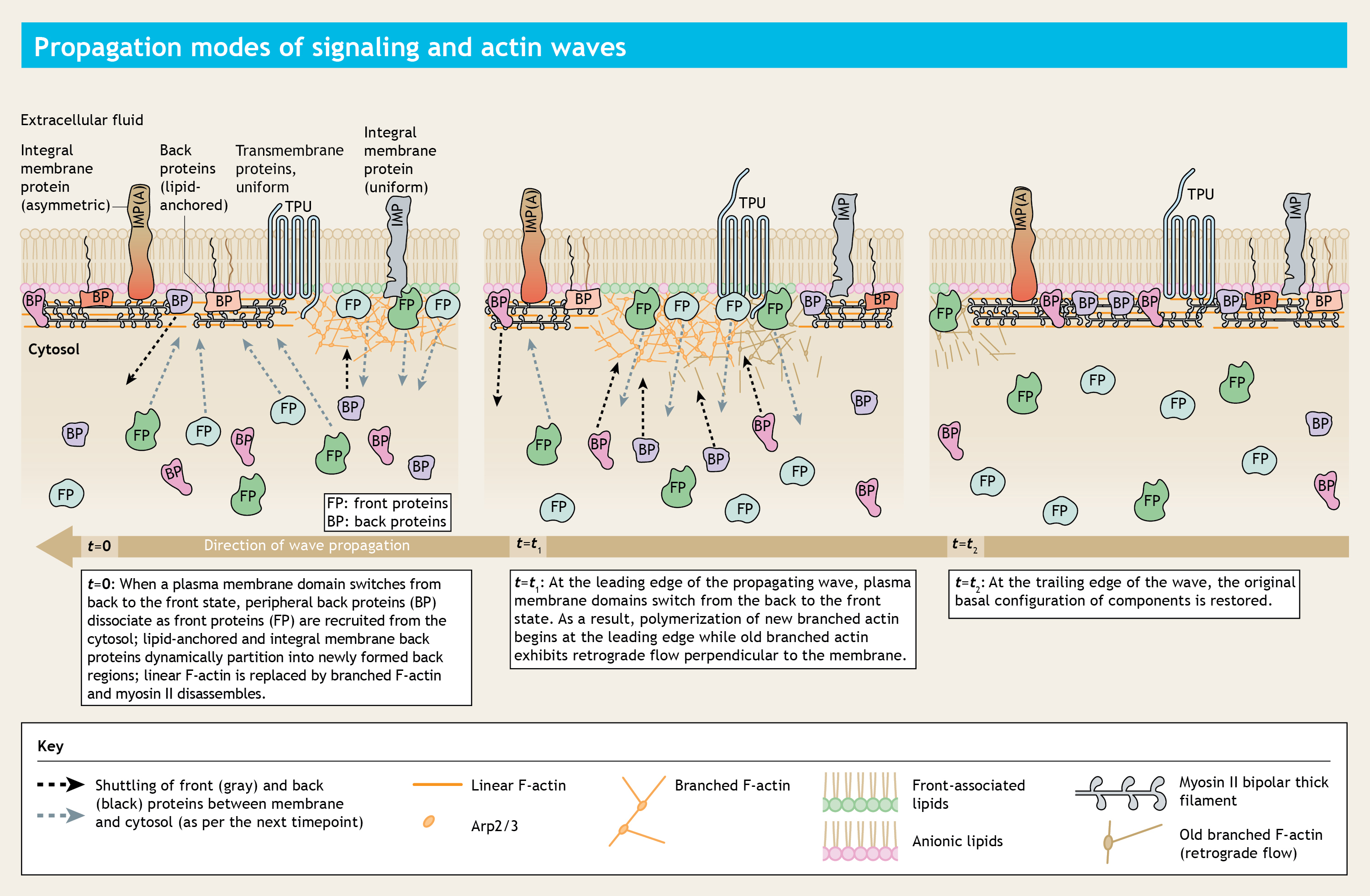
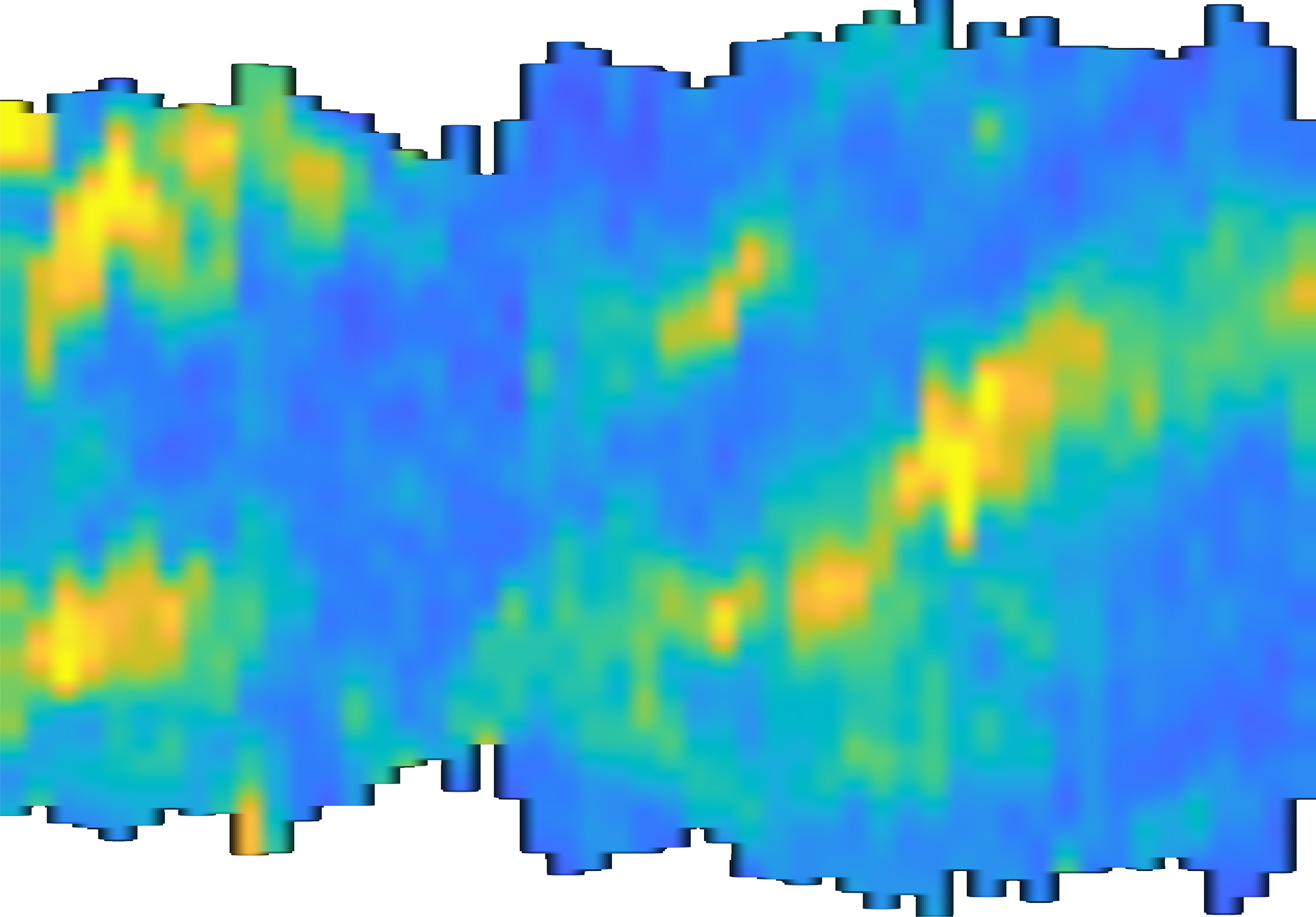
A time-lapsed representation of a plasma membrane where excitable waves of biochemical activity are propagating from right to left. Time is increasing from left to right.
To learn more about my research, please visit the publications page. Currently, I am developing next-generation of CAR-T cell therapies by identifying the design principles of immunological synapses. I am also working to understand how extreme environmental stresses alters cell physiologies. Please feel free to contact me for any questions or set up a discussion.